UV light has been used in medicine for over a century, including early dermatology treatments using phototherapy to treat skin conditions recognized with the 1903 Nobel Prize. While both Solius and traditional phototherapy devices use UVB light, they operate at different parts of the UVB spectrum.
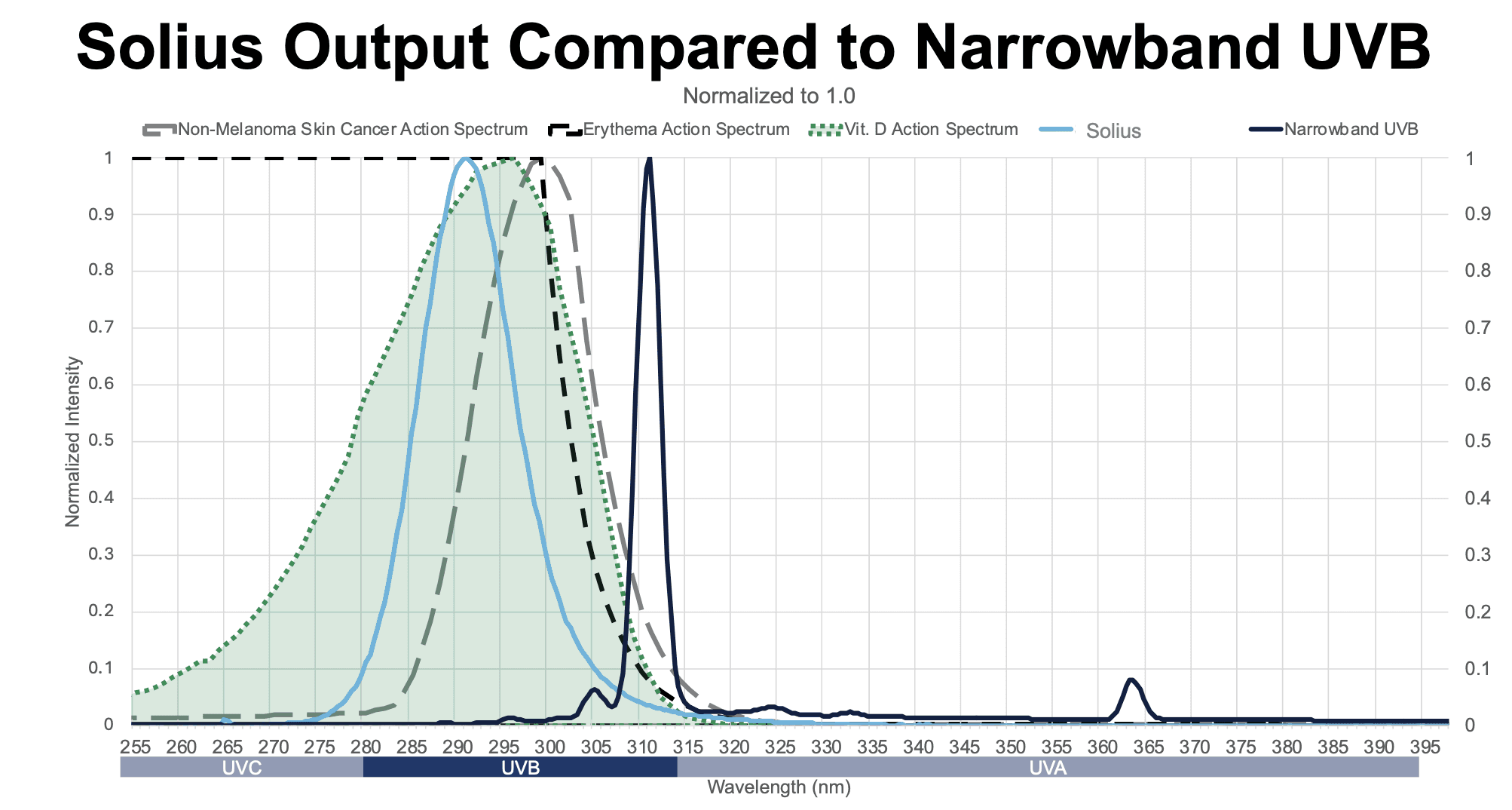
Narrowband UVB phototherapy is primarily used to treat chronic skin conditions such as psoriasis, eczema, and vitiligo, leveraging UVB bulb technology developed in the 1980s that emits a peak wavelength at 311–313 nm. While referred to as “narrowband,” these bulbs still produce secondary emissions outside the intended range, including harmful UVA emissions.
These treatments expose localized areas of affected skin to UV light, working by suppressing local immune activity and slowing abnormal skin cell growth. They are typically administered under medical supervision, with dosing carefully managed by a trained clinician based on a patient’s condition and skin type. At-home devices at this wavelength are generally prescription-based and intended for specific dermatological use. The 311-113 nm wavelength is less powerful than the Solius wavelength but was previously the best available option to balance safety and efficacy.
Solius is designed to work beyond the skin to safely deliver the beneficial biological effects of sunlight, including vitamin D₃ production and other light-driven pathways linked to metabolic, hormonal, microbiome, and immune health to drive systemic benefits.
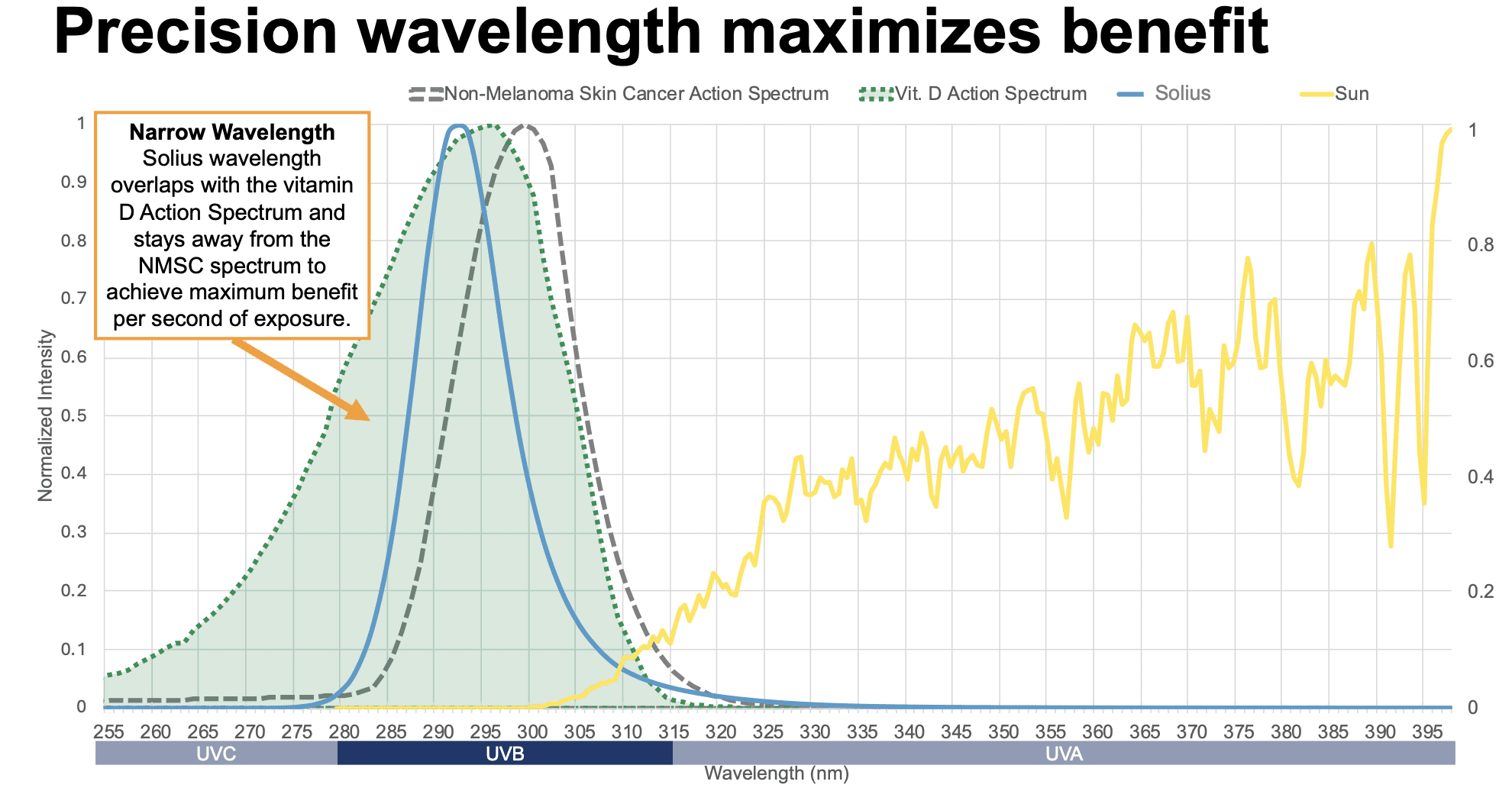
Decades of research show that the body produces vitamin D most efficiently in a specific portion of the UVB range, roughly between 290–315 nm, with peak effectiveness around 295–297 nm. Solius is tuned to ~293 nm, placing it close to this biological sweet spot and closer than traditional phototherapy devices. Within the UVB spectrum, even small changes in wavelength can significantly influence biological effects.
Solius is also positioned slightly below wavelengths more strongly associated with UV-induced DNA damage, which tend to peak closer to ~299–300 nm. This spectral positioning is designed to increase biological effectiveness while reducing overlap with higher-risk wavelengths.
Unlike traditional phototherapy systems, Solius incorporates an integrated safety and dosing system. This includes personalized dosing based on a patented and FDA-cleared skin sensor, automatic dose adjustments based on skin response, individual mobile app profiles for each user, therapy response feedback questions, distance sensing to ensure proper positioning, AI-assisted protective eyewear detection for eye safety, and enforced 24-hour lockout intervals between sessions to prevent overexposure.
Solius is the first FDA-cleared UVB device for over-the-counter use in this category, reflecting its focus on safe, consistent use outside of a clinical setting without a prescription.